|
11/8/2023 0 Comments Dapagliflozin declare trial 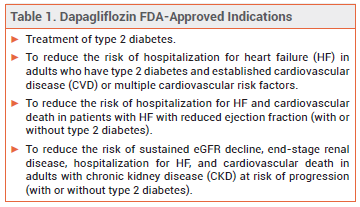
Any patient currently receiving chronic (>30 consecutive days) treatment with an oral steroid at a dose equivalent to oral prednisolone ≥10 mg (e.g., betamethasone ≥1.2 mg, dexamethasone ≥1.5 mg, hydrocortisone ≥40 mg) per day.Previous treatment with any SGLT2 inhibitor.Current or recent (within 12 months) treatment with rosiglitazone.Current or recent (within 24 months) treatment with pioglitazone and/or use of pioglitazone for 2 years or more at any time.Use of the following excluded medications: If at the time of enrollment, it is known that the patient will not meet criteria after a successful run-in period he/she should not be entered into run in. Patients should not meet any exclusion criteria at the time of randomization. For inclusion in the optional genetic research, patients must fulfill the criterion specified in WOCBP must be willing to use a medically accepted method of contraception that is considered reliable in the judgment of the Investigator.WOCBP include any female who has experienced menarche and who has not undergone successful surgical sterilization (hysterectomy, bilateral tubal ligation or bilateral oophorectomy) or is not postmenopausal. WOCBP must have a negative urine pregnancy test.WOCBP must take precautions to avoid pregnancy throughout the study and for 4 weeks after intake of the last dose. Age > 55 years in men and > 60 in women AND presence of at least 1 of the following additional risk factors (see Appendix E for details).Established CV Disease (See Appendix E for details) OR No known cardiovascular disease AND at least two cardiovascular risk factors in addition to High Risk for CV event defined as having either established CV disease and/or multiple risk factors: Provision of informed consent prior to any study specific procedures (including run-in).For Optum: Jan 8, 2014-(end of data availability).For Marketscan and Medicare: Jan 8, 2014-(end of data availability).Market availability of dapagliflozin in the U.S. Please see: for full code and algorithm definitions. Investigators assume that the RCT provides the reference standard treatment effect estimate and that failure to replicate RCT findings is indicative of the inadequacy of the healthcare claims data for replication for a range of possible reasons and does not provide information on the validity of the original RCT finding. Randomization is also not replicable in healthcare claims data but was proxied through a statistical balancing of measured covariates according to standard practice. Although many features of the trial cannot be directly replicated in healthcare claims, key design features, including outcomes, exposures, and inclusion/exclusion criteria, were selected to proxy those features from the trial. It is intended to replicate, as closely as is possible in healthcare insurance claims data, the trial listed below/above. This is a non-randomized, non-interventional study that is part of the RCT DUPLICATE initiative (of the Brigham and Women's Hospital, Harvard Medical School. Why Should I Register and Submit Results?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
